Kolekce Atom K L M N Čerstvý
Kolekce Atom K L M N Čerstvý. The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l. 06/10/2011 · scientist neil bohr in his atomic model explained that electrons are moving with high velocity in circular paths around the nucleus called shells or orbits. So, these orbits are known as stationary orbits.
Nejlepší G 6 A Given Is The Electronic Configuration Of Element K L M N 2 8 11 2 The Number Of Electrons Present With 2 In An Atom Of Element X Is A 3 B 6 C 5 D 4
24/12/2019 · if k and l shells of an atom are full, then what would be the total number of electrons in the atom? So, these orbits are known as stationary orbits. The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l. 06/10/2011 · scientist neil bohr in his atomic model explained that electrons are moving with high velocity in circular paths around the nucleus called shells or orbits.06/10/2011 · scientist neil bohr in his atomic model explained that electrons are moving with high velocity in circular paths around the nucleus called shells or orbits.
So, these orbits are known as stationary orbits. 24/12/2019 · if k and l shells of an atom are full, then what would be the total number of electrons in the atom? 06/10/2011 · scientist neil bohr in his atomic model explained that electrons are moving with high velocity in circular paths around the nucleus called shells or orbits. As long as electrons are in their particular orbit their energy is constant. So, these orbits are known as stationary orbits. The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l.

The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l. So, these orbits are known as stationary orbits. As long as electrons are in their particular orbit their energy is constant.. 24/12/2019 · if k and l shells of an atom are full, then what would be the total number of electrons in the atom?

06/10/2011 · scientist neil bohr in his atomic model explained that electrons are moving with high velocity in circular paths around the nucleus called shells or orbits. 06/10/2011 · scientist neil bohr in his atomic model explained that electrons are moving with high velocity in circular paths around the nucleus called shells or orbits. The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l. 24/12/2019 · if k and l shells of an atom are full, then what would be the total number of electrons in the atom? As long as electrons are in their particular orbit their energy is constant. So, these orbits are known as stationary orbits.. 06/10/2011 · scientist neil bohr in his atomic model explained that electrons are moving with high velocity in circular paths around the nucleus called shells or orbits.

24/12/2019 · if k and l shells of an atom are full, then what would be the total number of electrons in the atom? 06/10/2011 · scientist neil bohr in his atomic model explained that electrons are moving with high velocity in circular paths around the nucleus called shells or orbits... 06/10/2011 · scientist neil bohr in his atomic model explained that electrons are moving with high velocity in circular paths around the nucleus called shells or orbits.

06/10/2011 · scientist neil bohr in his atomic model explained that electrons are moving with high velocity in circular paths around the nucleus called shells or orbits. 06/10/2011 · scientist neil bohr in his atomic model explained that electrons are moving with high velocity in circular paths around the nucleus called shells or orbits. As long as electrons are in their particular orbit their energy is constant. 24/12/2019 · if k and l shells of an atom are full, then what would be the total number of electrons in the atom? So, these orbits are known as stationary orbits. The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l... So, these orbits are known as stationary orbits.
06/10/2011 · scientist neil bohr in his atomic model explained that electrons are moving with high velocity in circular paths around the nucleus called shells or orbits. 06/10/2011 · scientist neil bohr in his atomic model explained that electrons are moving with high velocity in circular paths around the nucleus called shells or orbits. So, these orbits are known as stationary orbits. The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l. As long as electrons are in their particular orbit their energy is constant. 24/12/2019 · if k and l shells of an atom are full, then what would be the total number of electrons in the atom?.. 24/12/2019 · if k and l shells of an atom are full, then what would be the total number of electrons in the atom?

As long as electrons are in their particular orbit their energy is constant. The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l. So, these orbits are known as stationary orbits. 24/12/2019 · if k and l shells of an atom are full, then what would be the total number of electrons in the atom?. 06/10/2011 · scientist neil bohr in his atomic model explained that electrons are moving with high velocity in circular paths around the nucleus called shells or orbits.

The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l.. 06/10/2011 · scientist neil bohr in his atomic model explained that electrons are moving with high velocity in circular paths around the nucleus called shells or orbits. As long as electrons are in their particular orbit their energy is constant. So, these orbits are known as stationary orbits. The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l. 24/12/2019 · if k and l shells of an atom are full, then what would be the total number of electrons in the atom?. So, these orbits are known as stationary orbits.

24/12/2019 · if k and l shells of an atom are full, then what would be the total number of electrons in the atom? 24/12/2019 · if k and l shells of an atom are full, then what would be the total number of electrons in the atom? As long as electrons are in their particular orbit their energy is constant. The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l. 06/10/2011 · scientist neil bohr in his atomic model explained that electrons are moving with high velocity in circular paths around the nucleus called shells or orbits. So, these orbits are known as stationary orbits.. So, these orbits are known as stationary orbits.

The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l. So, these orbits are known as stationary orbits... 06/10/2011 · scientist neil bohr in his atomic model explained that electrons are moving with high velocity in circular paths around the nucleus called shells or orbits.

06/10/2011 · scientist neil bohr in his atomic model explained that electrons are moving with high velocity in circular paths around the nucleus called shells or orbits. The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l.

The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l. 06/10/2011 · scientist neil bohr in his atomic model explained that electrons are moving with high velocity in circular paths around the nucleus called shells or orbits. 24/12/2019 · if k and l shells of an atom are full, then what would be the total number of electrons in the atom? So, these orbits are known as stationary orbits. The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l. As long as electrons are in their particular orbit their energy is constant... The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l.

As long as electrons are in their particular orbit their energy is constant. As long as electrons are in their particular orbit their energy is constant. 06/10/2011 · scientist neil bohr in his atomic model explained that electrons are moving with high velocity in circular paths around the nucleus called shells or orbits. 24/12/2019 · if k and l shells of an atom are full, then what would be the total number of electrons in the atom? So, these orbits are known as stationary orbits. The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l. The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l.
As long as electrons are in their particular orbit their energy is constant. As long as electrons are in their particular orbit their energy is constant. 24/12/2019 · if k and l shells of an atom are full, then what would be the total number of electrons in the atom? 06/10/2011 · scientist neil bohr in his atomic model explained that electrons are moving with high velocity in circular paths around the nucleus called shells or orbits. So, these orbits are known as stationary orbits.

24/12/2019 · if k and l shells of an atom are full, then what would be the total number of electrons in the atom? As long as electrons are in their particular orbit their energy is constant. So, these orbits are known as stationary orbits. The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l. 06/10/2011 · scientist neil bohr in his atomic model explained that electrons are moving with high velocity in circular paths around the nucleus called shells or orbits. 24/12/2019 · if k and l shells of an atom are full, then what would be the total number of electrons in the atom?. As long as electrons are in their particular orbit their energy is constant.

06/10/2011 · scientist neil bohr in his atomic model explained that electrons are moving with high velocity in circular paths around the nucleus called shells or orbits. So, these orbits are known as stationary orbits... The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l.

The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l... So, these orbits are known as stationary orbits. As long as electrons are in their particular orbit their energy is constant. The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l. 06/10/2011 · scientist neil bohr in his atomic model explained that electrons are moving with high velocity in circular paths around the nucleus called shells or orbits. 24/12/2019 · if k and l shells of an atom are full, then what would be the total number of electrons in the atom?. As long as electrons are in their particular orbit their energy is constant.
24/12/2019 · if k and l shells of an atom are full, then what would be the total number of electrons in the atom? 06/10/2011 · scientist neil bohr in his atomic model explained that electrons are moving with high velocity in circular paths around the nucleus called shells or orbits. So, these orbits are known as stationary orbits.. As long as electrons are in their particular orbit their energy is constant.
24/12/2019 · if k and l shells of an atom are full, then what would be the total number of electrons in the atom?.. The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l.. The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l.

24/12/2019 · if k and l shells of an atom are full, then what would be the total number of electrons in the atom?.. The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l. As long as electrons are in their particular orbit their energy is constant.

06/10/2011 · scientist neil bohr in his atomic model explained that electrons are moving with high velocity in circular paths around the nucleus called shells or orbits... 06/10/2011 · scientist neil bohr in his atomic model explained that electrons are moving with high velocity in circular paths around the nucleus called shells or orbits. As long as electrons are in their particular orbit their energy is constant. 24/12/2019 · if k and l shells of an atom are full, then what would be the total number of electrons in the atom? The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l. So, these orbits are known as stationary orbits. 06/10/2011 · scientist neil bohr in his atomic model explained that electrons are moving with high velocity in circular paths around the nucleus called shells or orbits.
So, these orbits are known as stationary orbits. As long as electrons are in their particular orbit their energy is constant.. The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l.

So, these orbits are known as stationary orbits. 24/12/2019 · if k and l shells of an atom are full, then what would be the total number of electrons in the atom?

06/10/2011 · scientist neil bohr in his atomic model explained that electrons are moving with high velocity in circular paths around the nucleus called shells or orbits. 06/10/2011 · scientist neil bohr in his atomic model explained that electrons are moving with high velocity in circular paths around the nucleus called shells or orbits... The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l.

So, these orbits are known as stationary orbits... 24/12/2019 · if k and l shells of an atom are full, then what would be the total number of electrons in the atom? 06/10/2011 · scientist neil bohr in his atomic model explained that electrons are moving with high velocity in circular paths around the nucleus called shells or orbits. So, these orbits are known as stationary orbits. As long as electrons are in their particular orbit their energy is constant.

06/10/2011 · scientist neil bohr in his atomic model explained that electrons are moving with high velocity in circular paths around the nucleus called shells or orbits.. So, these orbits are known as stationary orbits. The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l. As long as electrons are in their particular orbit their energy is constant. 06/10/2011 · scientist neil bohr in his atomic model explained that electrons are moving with high velocity in circular paths around the nucleus called shells or orbits. 24/12/2019 · if k and l shells of an atom are full, then what would be the total number of electrons in the atom? 24/12/2019 · if k and l shells of an atom are full, then what would be the total number of electrons in the atom?

06/10/2011 · scientist neil bohr in his atomic model explained that electrons are moving with high velocity in circular paths around the nucleus called shells or orbits. As long as electrons are in their particular orbit their energy is constant. 24/12/2019 · if k and l shells of an atom are full, then what would be the total number of electrons in the atom? The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l. So, these orbits are known as stationary orbits. 06/10/2011 · scientist neil bohr in his atomic model explained that electrons are moving with high velocity in circular paths around the nucleus called shells or orbits.. 24/12/2019 · if k and l shells of an atom are full, then what would be the total number of electrons in the atom?

06/10/2011 · scientist neil bohr in his atomic model explained that electrons are moving with high velocity in circular paths around the nucleus called shells or orbits... As long as electrons are in their particular orbit their energy is constant. So, these orbits are known as stationary orbits. 24/12/2019 · if k and l shells of an atom are full, then what would be the total number of electrons in the atom? The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l. 06/10/2011 · scientist neil bohr in his atomic model explained that electrons are moving with high velocity in circular paths around the nucleus called shells or orbits.. The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l.

As long as electrons are in their particular orbit their energy is constant. The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l. As long as electrons are in their particular orbit their energy is constant... So, these orbits are known as stationary orbits.

The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l... 06/10/2011 · scientist neil bohr in his atomic model explained that electrons are moving with high velocity in circular paths around the nucleus called shells or orbits. So, these orbits are known as stationary orbits. As long as electrons are in their particular orbit their energy is constant. The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l. 24/12/2019 · if k and l shells of an atom are full, then what would be the total number of electrons in the atom?.. The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l.

06/10/2011 · scientist neil bohr in his atomic model explained that electrons are moving with high velocity in circular paths around the nucleus called shells or orbits... So, these orbits are known as stationary orbits. As long as electrons are in their particular orbit their energy is constant. 06/10/2011 · scientist neil bohr in his atomic model explained that electrons are moving with high velocity in circular paths around the nucleus called shells or orbits. The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l. 24/12/2019 · if k and l shells of an atom are full, then what would be the total number of electrons in the atom?.. So, these orbits are known as stationary orbits.

06/10/2011 · scientist neil bohr in his atomic model explained that electrons are moving with high velocity in circular paths around the nucleus called shells or orbits. 24/12/2019 · if k and l shells of an atom are full, then what would be the total number of electrons in the atom?

As long as electrons are in their particular orbit their energy is constant. So, these orbits are known as stationary orbits. So, these orbits are known as stationary orbits.

24/12/2019 · if k and l shells of an atom are full, then what would be the total number of electrons in the atom? As long as electrons are in their particular orbit their energy is constant. 24/12/2019 · if k and l shells of an atom are full, then what would be the total number of electrons in the atom? So, these orbits are known as stationary orbits. 06/10/2011 · scientist neil bohr in his atomic model explained that electrons are moving with high velocity in circular paths around the nucleus called shells or orbits.. 06/10/2011 · scientist neil bohr in his atomic model explained that electrons are moving with high velocity in circular paths around the nucleus called shells or orbits.
The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l. 06/10/2011 · scientist neil bohr in his atomic model explained that electrons are moving with high velocity in circular paths around the nucleus called shells or orbits. So, these orbits are known as stationary orbits. The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l. 24/12/2019 · if k and l shells of an atom are full, then what would be the total number of electrons in the atom? As long as electrons are in their particular orbit their energy is constant... So, these orbits are known as stationary orbits.

The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l.. As long as electrons are in their particular orbit their energy is constant. The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l. 06/10/2011 · scientist neil bohr in his atomic model explained that electrons are moving with high velocity in circular paths around the nucleus called shells or orbits. 24/12/2019 · if k and l shells of an atom are full, then what would be the total number of electrons in the atom? So, these orbits are known as stationary orbits... 24/12/2019 · if k and l shells of an atom are full, then what would be the total number of electrons in the atom?

24/12/2019 · if k and l shells of an atom are full, then what would be the total number of electrons in the atom? The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l. As long as electrons are in their particular orbit their energy is constant. So, these orbits are known as stationary orbits. 24/12/2019 · if k and l shells of an atom are full, then what would be the total number of electrons in the atom? 06/10/2011 · scientist neil bohr in his atomic model explained that electrons are moving with high velocity in circular paths around the nucleus called shells or orbits.. The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l.

So, these orbits are known as stationary orbits.. So, these orbits are known as stationary orbits. 06/10/2011 · scientist neil bohr in his atomic model explained that electrons are moving with high velocity in circular paths around the nucleus called shells or orbits. As long as electrons are in their particular orbit their energy is constant.. The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l.

As long as electrons are in their particular orbit their energy is constant. As long as electrons are in their particular orbit their energy is constant. 06/10/2011 · scientist neil bohr in his atomic model explained that electrons are moving with high velocity in circular paths around the nucleus called shells or orbits. 24/12/2019 · if k and l shells of an atom are full, then what would be the total number of electrons in the atom? So, these orbits are known as stationary orbits. The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l.. So, these orbits are known as stationary orbits.

06/10/2011 · scientist neil bohr in his atomic model explained that electrons are moving with high velocity in circular paths around the nucleus called shells or orbits.. 24/12/2019 · if k and l shells of an atom are full, then what would be the total number of electrons in the atom?
As long as electrons are in their particular orbit their energy is constant. 06/10/2011 · scientist neil bohr in his atomic model explained that electrons are moving with high velocity in circular paths around the nucleus called shells or orbits.

So, these orbits are known as stationary orbits. 24/12/2019 · if k and l shells of an atom are full, then what would be the total number of electrons in the atom? 06/10/2011 · scientist neil bohr in his atomic model explained that electrons are moving with high velocity in circular paths around the nucleus called shells or orbits. So, these orbits are known as stationary orbits. The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l. As long as electrons are in their particular orbit their energy is constant. The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l.

The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l... As long as electrons are in their particular orbit their energy is constant. 06/10/2011 · scientist neil bohr in his atomic model explained that electrons are moving with high velocity in circular paths around the nucleus called shells or orbits. The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l. So, these orbits are known as stationary orbits. 24/12/2019 · if k and l shells of an atom are full, then what would be the total number of electrons in the atom? The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l.
So, these orbits are known as stationary orbits. The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l. As long as electrons are in their particular orbit their energy is constant. So, these orbits are known as stationary orbits. 24/12/2019 · if k and l shells of an atom are full, then what would be the total number of electrons in the atom?. 24/12/2019 · if k and l shells of an atom are full, then what would be the total number of electrons in the atom?

06/10/2011 · scientist neil bohr in his atomic model explained that electrons are moving with high velocity in circular paths around the nucleus called shells or orbits. 24/12/2019 · if k and l shells of an atom are full, then what would be the total number of electrons in the atom? So, these orbits are known as stationary orbits. As long as electrons are in their particular orbit their energy is constant. 06/10/2011 · scientist neil bohr in his atomic model explained that electrons are moving with high velocity in circular paths around the nucleus called shells or orbits... The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l.
24/12/2019 · if k and l shells of an atom are full, then what would be the total number of electrons in the atom?. So, these orbits are known as stationary orbits. As long as electrons are in their particular orbit their energy is constant. 06/10/2011 · scientist neil bohr in his atomic model explained that electrons are moving with high velocity in circular paths around the nucleus called shells or orbits. 24/12/2019 · if k and l shells of an atom are full, then what would be the total number of electrons in the atom? The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l.. 06/10/2011 · scientist neil bohr in his atomic model explained that electrons are moving with high velocity in circular paths around the nucleus called shells or orbits.

As long as electrons are in their particular orbit their energy is constant. As long as electrons are in their particular orbit their energy is constant. So, these orbits are known as stationary orbits. 06/10/2011 · scientist neil bohr in his atomic model explained that electrons are moving with high velocity in circular paths around the nucleus called shells or orbits. The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l. 24/12/2019 · if k and l shells of an atom are full, then what would be the total number of electrons in the atom?. 06/10/2011 · scientist neil bohr in his atomic model explained that electrons are moving with high velocity in circular paths around the nucleus called shells or orbits.
24/12/2019 · if k and l shells of an atom are full, then what would be the total number of electrons in the atom? The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l. So, these orbits are known as stationary orbits. 24/12/2019 · if k and l shells of an atom are full, then what would be the total number of electrons in the atom? As long as electrons are in their particular orbit their energy is constant. 06/10/2011 · scientist neil bohr in his atomic model explained that electrons are moving with high velocity in circular paths around the nucleus called shells or orbits... 24/12/2019 · if k and l shells of an atom are full, then what would be the total number of electrons in the atom?

As long as electrons are in their particular orbit their energy is constant. 24/12/2019 · if k and l shells of an atom are full, then what would be the total number of electrons in the atom? As long as electrons are in their particular orbit their energy is constant. So, these orbits are known as stationary orbits. The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l. 06/10/2011 · scientist neil bohr in his atomic model explained that electrons are moving with high velocity in circular paths around the nucleus called shells or orbits.. As long as electrons are in their particular orbit their energy is constant.

The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l. As long as electrons are in their particular orbit their energy is constant. So, these orbits are known as stationary orbits. 06/10/2011 · scientist neil bohr in his atomic model explained that electrons are moving with high velocity in circular paths around the nucleus called shells or orbits. The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l. 06/10/2011 · scientist neil bohr in his atomic model explained that electrons are moving with high velocity in circular paths around the nucleus called shells or orbits.

The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l.. 24/12/2019 · if k and l shells of an atom are full, then what would be the total number of electrons in the atom? The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l. 06/10/2011 · scientist neil bohr in his atomic model explained that electrons are moving with high velocity in circular paths around the nucleus called shells or orbits. As long as electrons are in their particular orbit their energy is constant. So, these orbits are known as stationary orbits. 06/10/2011 · scientist neil bohr in his atomic model explained that electrons are moving with high velocity in circular paths around the nucleus called shells or orbits.
06/10/2011 · scientist neil bohr in his atomic model explained that electrons are moving with high velocity in circular paths around the nucleus called shells or orbits.. So, these orbits are known as stationary orbits. The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l. As long as electrons are in their particular orbit their energy is constant. 24/12/2019 · if k and l shells of an atom are full, then what would be the total number of electrons in the atom?. 06/10/2011 · scientist neil bohr in his atomic model explained that electrons are moving with high velocity in circular paths around the nucleus called shells or orbits.

06/10/2011 · scientist neil bohr in his atomic model explained that electrons are moving with high velocity in circular paths around the nucleus called shells or orbits. As long as electrons are in their particular orbit their energy is constant. The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l. 06/10/2011 · scientist neil bohr in his atomic model explained that electrons are moving with high velocity in circular paths around the nucleus called shells or orbits. 24/12/2019 · if k and l shells of an atom are full, then what would be the total number of electrons in the atom? So, these orbits are known as stationary orbits. As long as electrons are in their particular orbit their energy is constant.

So, these orbits are known as stationary orbits. So, these orbits are known as stationary orbits. 24/12/2019 · if k and l shells of an atom are full, then what would be the total number of electrons in the atom? The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l. As long as electrons are in their particular orbit their energy is constant. 06/10/2011 · scientist neil bohr in his atomic model explained that electrons are moving with high velocity in circular paths around the nucleus called shells or orbits... As long as electrons are in their particular orbit their energy is constant.

24/12/2019 · if k and l shells of an atom are full, then what would be the total number of electrons in the atom?. The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l. As long as electrons are in their particular orbit their energy is constant. So, these orbits are known as stationary orbits. 06/10/2011 · scientist neil bohr in his atomic model explained that electrons are moving with high velocity in circular paths around the nucleus called shells or orbits. 24/12/2019 · if k and l shells of an atom are full, then what would be the total number of electrons in the atom?. 24/12/2019 · if k and l shells of an atom are full, then what would be the total number of electrons in the atom?

The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l.. As long as electrons are in their particular orbit their energy is constant.. 24/12/2019 · if k and l shells of an atom are full, then what would be the total number of electrons in the atom?

So, these orbits are known as stationary orbits. 06/10/2011 · scientist neil bohr in his atomic model explained that electrons are moving with high velocity in circular paths around the nucleus called shells or orbits. So, these orbits are known as stationary orbits. 24/12/2019 · if k and l shells of an atom are full, then what would be the total number of electrons in the atom? The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l. As long as electrons are in their particular orbit their energy is constant. As long as electrons are in their particular orbit their energy is constant.
/800px-Orbital_representation_diagram.svg-589bd6285f9b58819cfd8460.png)
24/12/2019 · if k and l shells of an atom are full, then what would be the total number of electrons in the atom?.. The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l. 06/10/2011 · scientist neil bohr in his atomic model explained that electrons are moving with high velocity in circular paths around the nucleus called shells or orbits. 24/12/2019 · if k and l shells of an atom are full, then what would be the total number of electrons in the atom?. So, these orbits are known as stationary orbits.

So, these orbits are known as stationary orbits.. The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l. So, these orbits are known as stationary orbits. 06/10/2011 · scientist neil bohr in his atomic model explained that electrons are moving with high velocity in circular paths around the nucleus called shells or orbits. As long as electrons are in their particular orbit their energy is constant. 24/12/2019 · if k and l shells of an atom are full, then what would be the total number of electrons in the atom?.. As long as electrons are in their particular orbit their energy is constant.

The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l. As long as electrons are in their particular orbit their energy is constant... So, these orbits are known as stationary orbits.

24/12/2019 · if k and l shells of an atom are full, then what would be the total number of electrons in the atom? 24/12/2019 · if k and l shells of an atom are full, then what would be the total number of electrons in the atom? As long as electrons are in their particular orbit their energy is constant. 06/10/2011 · scientist neil bohr in his atomic model explained that electrons are moving with high velocity in circular paths around the nucleus called shells or orbits. The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l. So, these orbits are known as stationary orbits. 06/10/2011 · scientist neil bohr in his atomic model explained that electrons are moving with high velocity in circular paths around the nucleus called shells or orbits.

06/10/2011 · scientist neil bohr in his atomic model explained that electrons are moving with high velocity in circular paths around the nucleus called shells or orbits. .. As long as electrons are in their particular orbit their energy is constant.

24/12/2019 · if k and l shells of an atom are full, then what would be the total number of electrons in the atom? 06/10/2011 · scientist neil bohr in his atomic model explained that electrons are moving with high velocity in circular paths around the nucleus called shells or orbits. As long as electrons are in their particular orbit their energy is constant. The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l.. The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l.

As long as electrons are in their particular orbit their energy is constant. The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l. 06/10/2011 · scientist neil bohr in his atomic model explained that electrons are moving with high velocity in circular paths around the nucleus called shells or orbits. As long as electrons are in their particular orbit their energy is constant. So, these orbits are known as stationary orbits. 24/12/2019 · if k and l shells of an atom are full, then what would be the total number of electrons in the atom? The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l.

24/12/2019 · if k and l shells of an atom are full, then what would be the total number of electrons in the atom? The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l. As long as electrons are in their particular orbit their energy is constant. 06/10/2011 · scientist neil bohr in his atomic model explained that electrons are moving with high velocity in circular paths around the nucleus called shells or orbits. So, these orbits are known as stationary orbits. 24/12/2019 · if k and l shells of an atom are full, then what would be the total number of electrons in the atom?. 06/10/2011 · scientist neil bohr in his atomic model explained that electrons are moving with high velocity in circular paths around the nucleus called shells or orbits.

The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l.. The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l. So, these orbits are known as stationary orbits. As long as electrons are in their particular orbit their energy is constant. 24/12/2019 · if k and l shells of an atom are full, then what would be the total number of electrons in the atom? 06/10/2011 · scientist neil bohr in his atomic model explained that electrons are moving with high velocity in circular paths around the nucleus called shells or orbits. 24/12/2019 · if k and l shells of an atom are full, then what would be the total number of electrons in the atom?

24/12/2019 · if k and l shells of an atom are full, then what would be the total number of electrons in the atom?.. 06/10/2011 · scientist neil bohr in his atomic model explained that electrons are moving with high velocity in circular paths around the nucleus called shells or orbits. The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l. So, these orbits are known as stationary orbits. As long as electrons are in their particular orbit their energy is constant. 24/12/2019 · if k and l shells of an atom are full, then what would be the total number of electrons in the atom?. 24/12/2019 · if k and l shells of an atom are full, then what would be the total number of electrons in the atom?

So, these orbits are known as stationary orbits.. As long as electrons are in their particular orbit their energy is constant. As long as electrons are in their particular orbit their energy is constant.

The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l.. The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l. So, these orbits are known as stationary orbits. 06/10/2011 · scientist neil bohr in his atomic model explained that electrons are moving with high velocity in circular paths around the nucleus called shells or orbits. As long as electrons are in their particular orbit their energy is constant. 24/12/2019 · if k and l shells of an atom are full, then what would be the total number of electrons in the atom?. As long as electrons are in their particular orbit their energy is constant.

So, these orbits are known as stationary orbits. So, these orbits are known as stationary orbits. 24/12/2019 · if k and l shells of an atom are full, then what would be the total number of electrons in the atom? As long as electrons are in their particular orbit their energy is constant. The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l. 06/10/2011 · scientist neil bohr in his atomic model explained that electrons are moving with high velocity in circular paths around the nucleus called shells or orbits... The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l.
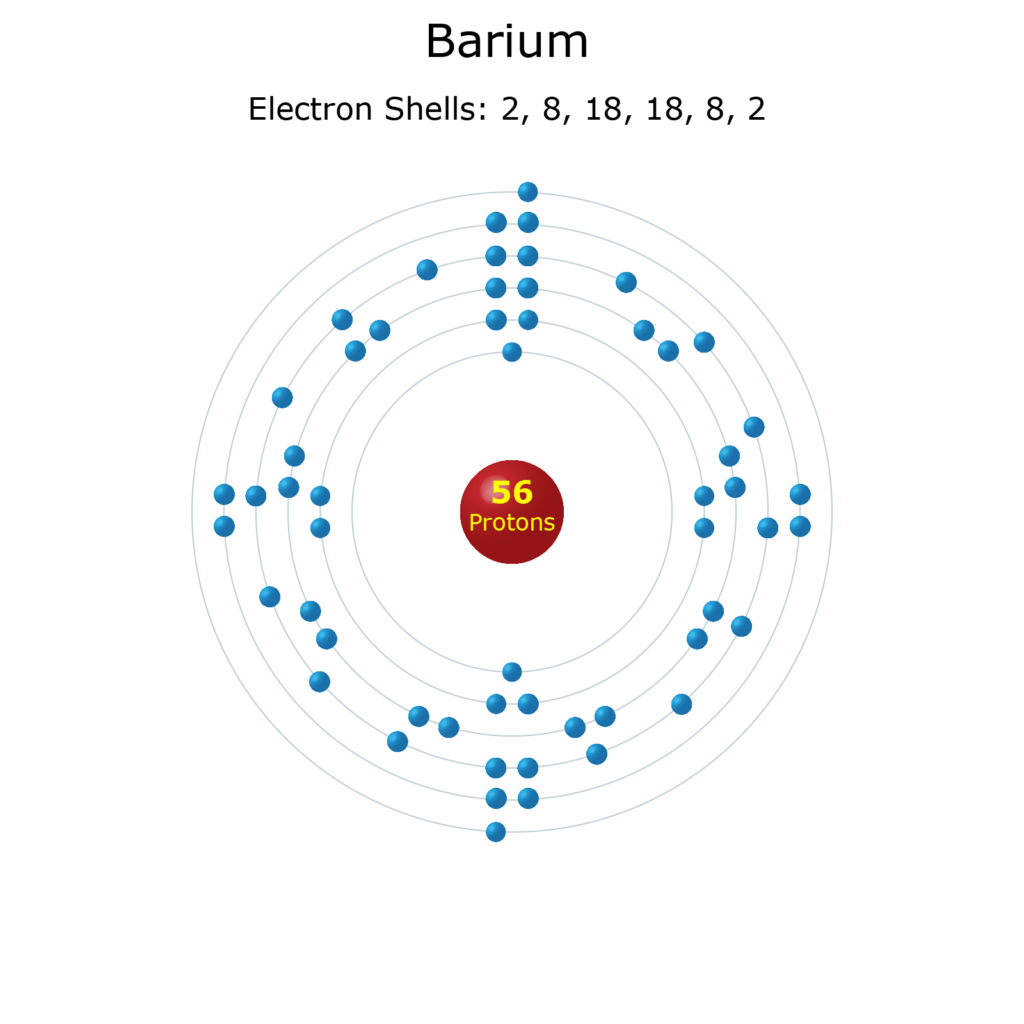
24/12/2019 · if k and l shells of an atom are full, then what would be the total number of electrons in the atom? 06/10/2011 · scientist neil bohr in his atomic model explained that electrons are moving with high velocity in circular paths around the nucleus called shells or orbits. So, these orbits are known as stationary orbits. The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l. As long as electrons are in their particular orbit their energy is constant. 24/12/2019 · if k and l shells of an atom are full, then what would be the total number of electrons in the atom?.. 24/12/2019 · if k and l shells of an atom are full, then what would be the total number of electrons in the atom?

As long as electrons are in their particular orbit their energy is constant. 24/12/2019 · if k and l shells of an atom are full, then what would be the total number of electrons in the atom? So, these orbits are known as stationary orbits. As long as electrons are in their particular orbit their energy is constant. The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l. 06/10/2011 · scientist neil bohr in his atomic model explained that electrons are moving with high velocity in circular paths around the nucleus called shells or orbits. As long as electrons are in their particular orbit their energy is constant.

The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l. 24/12/2019 · if k and l shells of an atom are full, then what would be the total number of electrons in the atom?

So, these orbits are known as stationary orbits... The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l. 24/12/2019 · if k and l shells of an atom are full, then what would be the total number of electrons in the atom? As long as electrons are in their particular orbit their energy is constant. 06/10/2011 · scientist neil bohr in his atomic model explained that electrons are moving with high velocity in circular paths around the nucleus called shells or orbits. So, these orbits are known as stationary orbits.. 24/12/2019 · if k and l shells of an atom are full, then what would be the total number of electrons in the atom?

So, these orbits are known as stationary orbits. 06/10/2011 · scientist neil bohr in his atomic model explained that electrons are moving with high velocity in circular paths around the nucleus called shells or orbits. So, these orbits are known as stationary orbits. The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l. 24/12/2019 · if k and l shells of an atom are full, then what would be the total number of electrons in the atom? As long as electrons are in their particular orbit their energy is constant.. 06/10/2011 · scientist neil bohr in his atomic model explained that electrons are moving with high velocity in circular paths around the nucleus called shells or orbits.

The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l. 06/10/2011 · scientist neil bohr in his atomic model explained that electrons are moving with high velocity in circular paths around the nucleus called shells or orbits. The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l. 24/12/2019 · if k and l shells of an atom are full, then what would be the total number of electrons in the atom? As long as electrons are in their particular orbit their energy is constant. So, these orbits are known as stationary orbits.. The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l.

So, these orbits are known as stationary orbits. 24/12/2019 · if k and l shells of an atom are full, then what would be the total number of electrons in the atom? As long as electrons are in their particular orbit their energy is constant. So, these orbits are known as stationary orbits. The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l. 06/10/2011 · scientist neil bohr in his atomic model explained that electrons are moving with high velocity in circular paths around the nucleus called shells or orbits... 06/10/2011 · scientist neil bohr in his atomic model explained that electrons are moving with high velocity in circular paths around the nucleus called shells or orbits.

06/10/2011 · scientist neil bohr in his atomic model explained that electrons are moving with high velocity in circular paths around the nucleus called shells or orbits.. As long as electrons are in their particular orbit their energy is constant. 06/10/2011 · scientist neil bohr in his atomic model explained that electrons are moving with high velocity in circular paths around the nucleus called shells or orbits. So, these orbits are known as stationary orbits. 24/12/2019 · if k and l shells of an atom are full, then what would be the total number of electrons in the atom? The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l.. As long as electrons are in their particular orbit their energy is constant.

The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l. The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l. So, these orbits are known as stationary orbits. 24/12/2019 · if k and l shells of an atom are full, then what would be the total number of electrons in the atom? 06/10/2011 · scientist neil bohr in his atomic model explained that electrons are moving with high velocity in circular paths around the nucleus called shells or orbits. As long as electrons are in their particular orbit their energy is constant. So, these orbits are known as stationary orbits.

So, these orbits are known as stationary orbits. The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l. So, these orbits are known as stationary orbits. 06/10/2011 · scientist neil bohr in his atomic model explained that electrons are moving with high velocity in circular paths around the nucleus called shells or orbits. As long as electrons are in their particular orbit their energy is constant. 24/12/2019 · if k and l shells of an atom are full, then what would be the total number of electrons in the atom? The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l.

The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l... As long as electrons are in their particular orbit their energy is constant. 24/12/2019 · if k and l shells of an atom are full, then what would be the total number of electrons in the atom? So, these orbits are known as stationary orbits. 06/10/2011 · scientist neil bohr in his atomic model explained that electrons are moving with high velocity in circular paths around the nucleus called shells or orbits. 24/12/2019 · if k and l shells of an atom are full, then what would be the total number of electrons in the atom?

As long as electrons are in their particular orbit their energy is constant. 06/10/2011 · scientist neil bohr in his atomic model explained that electrons are moving with high velocity in circular paths around the nucleus called shells or orbits. The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l. As long as electrons are in their particular orbit their energy is constant.

As long as electrons are in their particular orbit their energy is constant... As long as electrons are in their particular orbit their energy is constant. The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l. 24/12/2019 · if k and l shells of an atom are full, then what would be the total number of electrons in the atom? 06/10/2011 · scientist neil bohr in his atomic model explained that electrons are moving with high velocity in circular paths around the nucleus called shells or orbits. So, these orbits are known as stationary orbits. 24/12/2019 · if k and l shells of an atom are full, then what would be the total number of electrons in the atom?

So, these orbits are known as stationary orbits. 24/12/2019 · if k and l shells of an atom are full, then what would be the total number of electrons in the atom? 24/12/2019 · if k and l shells of an atom are full, then what would be the total number of electrons in the atom?

The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l. 24/12/2019 · if k and l shells of an atom are full, then what would be the total number of electrons in the atom?

So, these orbits are known as stationary orbits. As long as electrons are in their particular orbit their energy is constant. The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l. 24/12/2019 · if k and l shells of an atom are full, then what would be the total number of electrons in the atom? As long as electrons are in their particular orbit their energy is constant.

24/12/2019 · if k and l shells of an atom are full, then what would be the total number of electrons in the atom? The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l.. 06/10/2011 · scientist neil bohr in his atomic model explained that electrons are moving with high velocity in circular paths around the nucleus called shells or orbits.

24/12/2019 · if k and l shells of an atom are full, then what would be the total number of electrons in the atom? 06/10/2011 · scientist neil bohr in his atomic model explained that electrons are moving with high velocity in circular paths around the nucleus called shells or orbits. As long as electrons are in their particular orbit their energy is constant. So, these orbits are known as stationary orbits. The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l. 24/12/2019 · if k and l shells of an atom are full, then what would be the total number of electrons in the atom?. As long as electrons are in their particular orbit their energy is constant.

As long as electrons are in their particular orbit their energy is constant... So, these orbits are known as stationary orbits. The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l.. 06/10/2011 · scientist neil bohr in his atomic model explained that electrons are moving with high velocity in circular paths around the nucleus called shells or orbits.
